近年来,随着有色金属加工行业的迅猛发展,水体环境中重金属镍(Ni(Ⅱ))的污染已经成为人类面临的重要环境问题[1]。Ni(Ⅱ)对生物体具有致癌性[2]和致毒性[3],进入水体环境后形态稳定,难以被微生物所降解。工业生产中对于含Ni(Ⅱ)废水的传统处理方法包括化学沉淀法[4]、离子交换法[5]、膜分离法[6]、离子浮选法[7]等,它们往往成本高且处理过程复杂。相较而言,吸附法因其操作简便常作为去除Ni(Ⅱ)的一种有效方法运用在工业生产过程中[8],而如何制备高效率且成本低廉的吸附材料一直是众多学者的研究重点。在吸附材料中,炭质材料来源广泛且稳定性好[9],但其对于Ni(Ⅱ)的吸附量比较有限,而常用的煤又属于不可再生能源,成本较高。随着现代废物利用技术的发展,利用农林废弃物作为可持续替代资源制备炭质材料[10],并对其进行改性[11],改善其吸附性能,对于实现“以废治废”具有重大的现实意义。
目前,关于炭质材料制备条件的优化主要是采用单因素统计分析法和正交优化法[12-13],前者在试验组分复杂、试验因素较多的情况下所需试验次数多,试验周期长,且往往无法达到预期的效果;后者虽然对试验次数进行了精简,但有时不能直接使用,需要结合如模糊数学理论等对其数据进行前处理,使用范围受到了限制[14]。响应面分析法能克服这些弊端,通过合理的试验设计,在较少的试验次数下,直接对试验进行全面的分析,并以多元二次回归方程拟合因素与响应值之间的函数关系来寻求最优的参数条件[15-16]。现阶段,利用响应面法优化炭质材料的制备条件是国内外学者的关注重点[17-18],但却鲜有报道将此法应用到优化以农林废弃物为原料制备炭质材料,并对其进行改性去除水中Ni(Ⅱ)的过程当中。基于此,本研究采用以来源丰富且现有资源浪费严重的花生壳为原材料自制花生壳炭,并用价格低廉的高锰酸钾和氢氧化钾对其进行改性。利用Box-Behnken中心组合设计原理,采用响应面法探究花生壳炭吸附Ni(Ⅱ)的最优改性条件,以期获得较大的镍吸附量,为含镍废水的处理与获得高效廉价且具有可持续发展潜力的炭质材料的应用提供支持。利用扫描电子显微镜、比表面积分析仪等对改性前后的花生壳炭进行表征,分析材料的结构特征和表面特性;通过傅里叶红外光谱分析方法初步探究吸附材料除Ni(Ⅱ)的作用机制。
1 材料与方法 1.1 材料花生壳炭:利用废弃的花生壳,采用慢速热解法于真空气氛箱式炉中进行烧制,得到的生物质炭经冷却后研磨过60目分样筛,储存备用。
1.2 方法 1.2.1 改性花生壳炭的制备称取花生壳炭,与一定质量浓度的高锰酸钾(KMnO4)溶液混合,室温下充分浸渍后置于60 ℃的干燥箱中干燥4 h。取出后加入一定质量的氢氧化钾(KOH),在一定的热处理温度下得到改性花生壳炭。经冷却后碾磨过60目分样筛,储存备用。
1.2.2 改性花生壳炭吸附Ni(Ⅱ)的方法准确量取50 mL初始质量浓度为100 mg/L的镍离子溶液于100 mL的锥形瓶中,然后称取并加入0.05 g的改性花生壳炭。在温度为25 ℃的恒温振荡器中,以160 r/min的条件恒温振荡24 h。吸附完成后,将悬浮液经0.45 μm的滤膜过滤,用分光光度计测定溶液中Ni(Ⅱ)的质量浓度。
1.2.3 Ni(Ⅱ)含量的测定溶液中的Ni(Ⅱ)采用丁二酮肟分光光度法[19]测定。
1.2.4 响应面实验设计在前期预实验的基础上选取对改性效果影响显著的3个因素(热处理温度、KOH与炭的质量比、KMnO4的质量浓度)进行考察,并采用响应面法对这3个因素进行优化,以Ni(Ⅱ)的吸附量为响应值,设计三因素三水平实验,对花生壳炭的改性制备条件进行优化。实验因素和水平见表 1。
| 表 1 响应面分析因素及水平 Table 1 Factors and levels in response surface design |
实验数据采用Design-Expert 8.0.6软件进行分析。响应面数据分析采用二项式模型如下:
| $Y{\rm{ = }}{\mathit{\beta }_{\rm{0}}} + \sum\limits_{i = 1}^4 {{\mathit{\beta }_\mathit{i}}{\mathit{x}_\mathit{i}}} + \sum\limits_{i = 1}^4 {{\mathit{\beta }_{\mathit{ii}}}x_i^2} + \sum\limits_{i = 1}^4 {\sum\limits_{j \ge i}^4 {{\mathit{\beta }_{\mathit{ij}}}{x_\mathit{i}}{\mathit{x}_\mathit{j}}} } , $ | (1) |
式中:Y为响应值,β0、βi、βii和βij分别表示常数项、线性项、平方项、二次交互项的系数,xi、xj表示自变量。最终模型的拟合度采用方差分析进行评价,并运用Design-Expert 8.0.6绘制出三维曲面和相应的等值线图。
1.2.5 表征测试方法采用扫描电子显微镜(SEM,上海禹重实业有限公司FEI Nova NanoSEM型)分析改性前后花生壳炭的表面形貌信息。利用BET法(美国Micromeritics公司ASAP2020型)和Zeta电位分析仪(马尔文仪器公司Zetasizer Nano ZS型)分别得到改性前后材料的比表面积、孔容、孔径以及Zeta电位的相关参数。对吸附Ni(Ⅱ)前后的改性花生壳炭进行傅里叶红外光谱(FTIR,Thermo仪器公司Nicolet iS5型)测定,分析其表面官能团的变化。
2 结果与讨论 2.1 响应面实验结果根据实验数据并利用式(1)计算出材料对Ni(Ⅱ)的吸附量,结果见表 2。
| 表 2 响应面实验设计方案及结果 Table 2 Experimental design for response surface design and corresponding experimental data |
采用Design Expert 8.0.6软件对实验数据进行回归分析,并得出影响因素的一次效应、二次效应及其交互效应的关联方程,对改性花生壳炭的制备条件进行优化,并作响应面图。多元回归拟合分析得到吸附量与各因素变量的二次方程模型如式(2)所示:Y(吸附量)=9.38A+0.813 8B+0.551 2C-2.71AB-1.69AC+
| $\begin{array}{l} Y\left( {{\rm{吸附量}}} \right) = 9.38\mathit{A }+{\rm{0}}{\rm{.813\;8}}\mathit{B }+{\rm{0}}{\rm{.551\;2}}\mathit{C }-{\rm{2}}{\rm{.71}}\mathit{AB }-{\rm{1}}{\rm{.69}}\mathit{AC }+\\ {\rm{\;\;\;\;\;\;\;\;\;\;\;\;\;\;}}0.367\;5\mathit{BC }-{\rm{20}}{\rm{.29}}{\mathit{A}^{\rm{2}}} - 3.76{B^2} - 1.74{\mathit{C}^{\rm{2}}}。\end{array} $ | (2) |
对回归方程进行方差分析,从表 3可以看出:建立的回归模型极显著(p<0.000 1),说明方程拟合度好;失拟项值为0.511 5(>0.05),说明失拟项并不显著,残差由于随机误差而引起,模型选择正确;相关系数R2为0.999 2,表明预测值与实验值之间具有较高的相关性;调整性决定系数RAdj2为0.998 2,表明方程模型可信度较高,可较好的描述本次试验的结果。同时自变量一次项A、C,二次项AB、AC、A2、B2对吸附量影响极显著(p<0.01)。一次项C对吸附量影响显著(p<0.05)。
| 表 3 方差分析表 Table 3 Analysis of variance |
在响应面分析中,将一个因素固定在零水平,对另外两个因素进行分析,从中得到各因素对响应值的影响。图 1表示在KMnO4的质量浓度不变(0.75%)的条件下,考察热处理温度和KOH与炭的质量比对Ni(Ⅱ)吸附量的影响。从图中可知,吸附量随着热处理温度和KOH与炭质量比的增大呈先增大后减小的趋势。这是因为温度与KOH质量分数的增高有利于改善花生壳炭的孔隙结构,增大比表面积[20],所以吸附量升高。但过高的温度会导致既成孔道的坍塌,-COOH、-OH等活性官能团的减少[21],从而导致吸附量的下降。
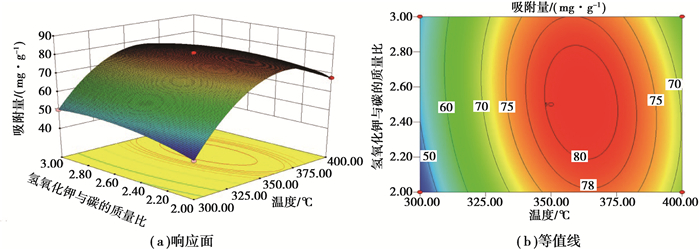
|
图 1 热处理温度和氢氧化钾与炭的质量比对Ni(Ⅱ)吸附量的影响:高锰酸钾质量浓度(75%) Figure 1 Effects of temperature and the mass ratio of KOH to carbon at a constant concentration of KMnO4(75%) on the adsorption capacity of Ni(Ⅱ) |
维持KOH与炭的质量比(2.5)不变,热处理温度和KMnO4质量浓度对Ni(Ⅱ)吸附量的影响如图 2所示。从图 2可以看出吸附量随着热处理温度和KMnO4质量浓度的增加呈先增大后减小的趋势。这是因为KMnO4在热解时产生氧气,对花生壳炭的造孔有促进作用[22];但随着KMnO4质量浓度的增加,反应变得越来越剧烈,大量的氧气对炭表面结构起到一定的破坏作用,使得原有孔道坍塌或者使产生的炭的碎渣堵塞孔隙,导致吸附量的下降[23]。
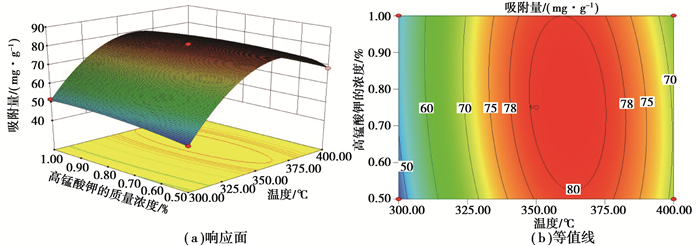
|
图 2 热处理温度与高锰酸钾的浓度对Ni(Ⅱ)吸附量的影响:氢氧化钾与炭的质量比(2.5) Figure 2 Effects of temperature and the concentration of KMnO4 at a constant mass ratio of KOH to carbon (2.5) on the adsorption capacity of Ni(Ⅱ) |
图 3表示在热处理温度(350 ℃)不变的条件下,考察KOH与炭的质量比和KMnO4质量浓度对Ni(Ⅱ)吸附量的影响。从图中可以看出当温度不变时,吸附量随着KOH与炭的质量比和KMnO4质量浓度的增加呈先增加后减小的趋势。相较温度对吸附量的大影响程度,KOH与KMnO4对吸附量的影响程度较小一些。结合响应面图,可知各因素对吸附量的显著性为热处理温度>KOH与炭的质量比>KMnO4质量浓度。
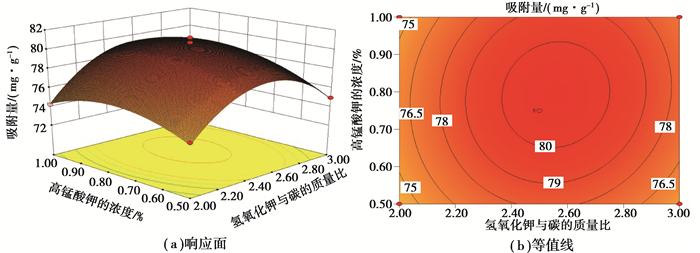
|
图 3 氢氧化钾与炭的质量比与高锰酸钾的浓度对Ni(Ⅱ)吸附量的影响:热处理温度(350 ℃) Figure 3 Effects of the constant mass ratio of KOH to carbon and the concentration of KMnO4 at a constant temperature (350 ℃) on the adsorption capacity of Ni(Ⅱ) |
根据模型可得制备改性花生壳炭的最优条件为:热处理温度361.348 ℃,KOH与炭的质量比为2.515,KMnO4的质量浓度为0.763%,在此条件下得到的理论吸附量为81.55 mg/g。为了检验预测结果是否与真实情况相一致,根据上述结果进行验证试验。考虑实际操作情况,将热处理温度修改为361 ℃,KOH与炭的质量比改为2.5,KMnO4的质量浓度改为0.76%,在此条件下进行试验得到的吸附量为85.02 mg/g,与理论值相近,说明优化结果可靠。在相同的吸附实验条件下,测得改性前花生壳炭对Ni(Ⅱ)的吸附量为5.45 mg/g,改性后的吸附量是改性前的15.6倍,极大的改善了其对水中Ni(Ⅱ)的吸附量。
2.4 改性花生壳炭SEM、BET及Zeta电位分析对改性前后的花生壳炭进行了SEM测试,结果见图 4。
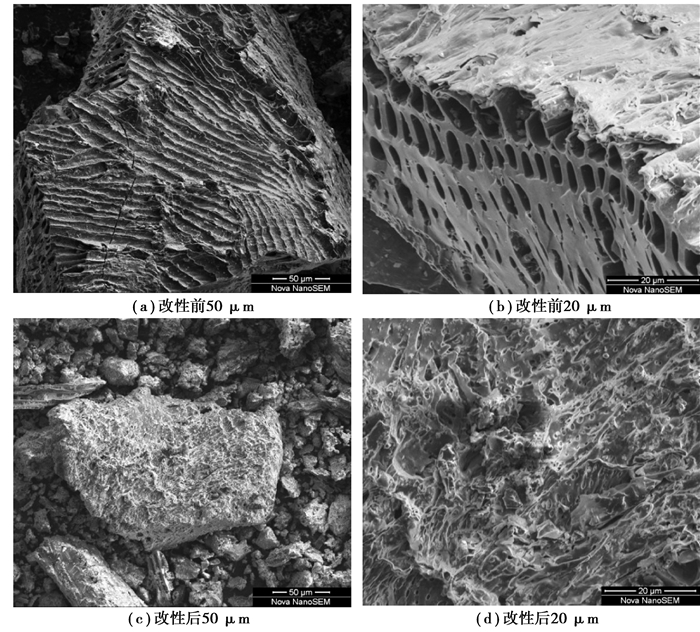
|
图 4 改性前后花生壳炭的电子扫描显微镜照片 Figure 4 SEM images of peanut shell carbon before and after modification |
从图 4中可以看出,改性前花生壳炭的表面较平整,依旧保留了原材料花生壳的部分形貌。纵向是花生壳炭表面的一些管状孔结构,将其进一步放大后(Fig. 4 (b)),发现其孔隙结构较疏松,表面几乎没有微孔。改性后,花生壳炭的表面形貌发生了较大的变化,表面变得粗糙,出现了一些微孔,材料的粒径相较改性前更小。这可能是因为改性过程反应比较剧烈,改性剂造孔作用明显,同时推测在改性后有更多的官能团修饰到了炭表面[23]。
吸附材料的比表面积、孔容和孔径是影响吸附效果的重要因素[24]。相关研究表明吸附材料的比表面积越大,提供的吸附位点越多,对重金属的吸附量就越大[25]。表 4是利用BET法计算得到的改性前后花生壳炭的比表面积、平均孔容和平均孔径的数据。从表中可得,改性后,花生壳炭的比表面积与平均孔容得到了极大的增加,平均孔径减小,更有利于对Ni(Ⅱ)的吸附。
| 表 4 改性前后花生壳炭的比表面特征参数 Table 4 BET parameters of peanut shell carbon before and after modification |
材料的Zeta电位可以反映物质的稳定性,当Zeta电位的绝对值越大,颗粒间的排斥也就越大,颗粒会分散得更均匀,稳定性就更好[26]。在中性条件下,测得改性前后花生壳炭的Zeta电位值分别为-34.3 mV、-41.1 mV,说明改性改善了花生壳炭的分散度与稳定性。
2.5 吸附反应机理为了进一步研究改性花生壳炭对Ni(Ⅱ)的吸附机理,利用FTIR对吸附前后的改性花生壳炭进行测定,测定结果如图 5所示。

|
图 5 改性花生壳炭吸附Ni(Ⅱ)前后的红外光谱图 Figure 5 FTIR spectra for modified peanut shell carbon before and after Ni(Ⅱ) adsorption |
图 5中3 445 cm-1为缔和羟基的伸缩振动峰,吸附Ni(Ⅱ)后强度减弱,1 020 cm-1处代表醇-CO-的吸收峰在吸附后红移至1 034 cm-1,表明改性花生壳炭表面的-OH在吸附中起作用[27-28],推断表面羟基在吸附Ni(Ⅱ)的过程中可能参与以下共沉淀反应和络合反应[21, 29]。
| $2{\rm{O}}{{\rm{H}}^ - } + {\rm{N}}{{\rm{i}}^{2 + }} \to {\rm{Ni}}\left( {{\rm{O}}{{\rm{H}}_{\rm{2}}}} \right) \downarrow , $ | (3) |
| ${\rm{R}} - {\rm{OH + N}}{{\rm{i}}^{2 + }} \to {\rm{R - O - Ni}}{\rm{。}}\ $ | (4) |
与此同时,代表- NH2面内弯曲振动吸收峰和伸缩振动峰的1 628 cm-1、1 568 cm-1在吸附了Ni(Ⅱ)后分别位移至1 626 cm-1、1558 cm-1,表明-NH2在吸附Ni(Ⅱ)的过程中同样起作用[30],此推断同Panda等的研究报道一致[31],推断-NH2参与的络合反应如下:
| $ - {\rm{N}}{{\rm{H}}_{\rm{2}}} + {\rm{N}}{{\rm{i}}^{{\rm{2 + }}}} \to - {\rm{N}}{{\rm{H}}_{\rm{2}}} - {\rm{Ni}}{\rm{。}} $ | (5) |
除了-OH、-NH2的反应外,吸附后代表芳香杂环的特征峰由于芳香环中C=C键角的拉伸,从874 cm-1蓝移至873 cm-1,推测改性花生壳炭芳香环中的π共轭芳香结构可作为电子供体与溶液中的Ni2+产生较弱的阳离子-π作用,从而将Ni2+吸附至炭材料的表面或孔中[32]。
3 结论笔者利用Box-Behnken中心组合设计实验,通过响应面法探究花生壳炭吸附水中镍离子的最优改性条件,采用SEM、BET等方法对改性前后的花生壳炭进行系统的表征,分析材料的结构特征与表面特性,并通过FTIR讨论改性花生壳炭的吸附机理,得到以下结论:
1) 通过响应面模型得到花生壳炭的最优改性条件为:热处理温度361 ℃,氢氧化钾与炭的质量比2.5,高锰酸钾的质量浓度0.76%。在该条件下制备的改性花生壳炭对Ni(Ⅱ)的吸附量为85.02 mg/g,是改性前的15.6倍,吸附性能得到较大改善,能够更高效的去除水中的重金属Ni(Ⅱ),具有很好的实用价值。
2) SEM结果显示改性后花生壳炭的粒径相较改性前变小,表面更为粗糙,出现微孔结构。BET结果表明改性后材料的比表面积和平均孔容增加,平均孔径减小,更有利于吸附水中的Ni(Ⅱ)。中性条件下测得改性后的花生壳炭Zeta电位的绝对值大于改性前,说明改性提高了材料的分散度与稳定性。
3) 对吸附反应前后的改性花生壳炭进行FTIR分析,结果表明-OH、-NH2是参与吸附反应的主要官能团,这些官能团与Ni(Ⅱ)发生共沉淀反应与络合反应。除此之外,改性花生壳炭芳香环中的π共轭芳香结构可以作为电子供体与溶液中的Ni2+产生较弱的阳离子-π作用,从而将Ni2+吸附至碳材料的表面或孔中。
本研究为废弃花生壳的利用开辟了一条新思路,同时利用响应面法优化得到了一种对水中镍离子具有良好吸附性能、成本较低的新型环保材料。
| [1] |
International Nickel Study Group. World Nickel Statistics[DB/OL].[2017-11-25] (2018-05-21) http://www.insg.org/prodnickel.aspx.
|
| [2] |
Li H X, Ji H B, Shi C J, et al. Distribution of heavy metals and metalloids in bulk and particle size fractions of soils from coal-mine brownfield and implications on human health[J]. Chemosphere, 2017, 172: 505-515. DOI:10.1016/j.chemosphere.2017.01.021 |
| [3] |
Gissi F, Stauber J L, Binet M T, et al. A review of nickel toxicity to marine and estuarine tropical biota with particular reference to the South East Asian and Melanesian region[J]. Environmental Pollution, 2016, 218: 1308-1323. DOI:10.1016/j.envpol.2016.08.089 |
| [4] |
Shih Y J, Lin C P, Huang Y H. Application of Fered-Fenton and chemical precipitation process for the treatment of electroless nickel plating wastewater[J]. Separation & Purification Technology, 2013, 104(4): 100-105. |
| [5] |
Moghbeli M R, Khajeh A, Alikhani M. Nanosilica reinforced ion-exchange polyHIPE type membrane for removal of nickel ions:Preparation, characterization and adsorption studies[J]. Chemical Engineering Journal, 2017, 309: 552-562. DOI:10.1016/j.cej.2016.10.048 |
| [6] |
Yurekli Y, Yildirim M, Aydin L, et al. Filtration and removal performances of membrane adsorbers[J]. Journal of Hazardous Materials, 2017, 332: 33-41. DOI:10.1016/j.jhazmat.2017.02.061 |
| [7] |
Farrokhpay S, Filippov L. Challenges in processing nickel laterite ores by flotation[J]. International Journal of Mineral Processing, 2016, 151: 59-67. DOI:10.1016/j.minpro.2016.04.007 |
| [8] |
Rashid A, Bhatti H N, Lqbal M, et al. Fungal biomass composite with bentonite efficiency for nickel and zinc adsorption:A mechanistic study[J]. Ecological Engineering, 2016, 91: 459-471. DOI:10.1016/j.ecoleng.2016.03.014 |
| [9] |
赵梦奇, 司马义·努尔拉, 米红宇. 基于多孔碳材料对重金属离子吸附性能的研究进展[J]. 材料科学与工程学报, 2014, 32(2): 301-306. ZHAO Mengqi, Ismayil Nurulla, MI Hongyu. Research progress on adsorption properties of porous carbon materials to heavy metal ions[J]. Journal of Materials Science & Engineering, 2014, 32(2): 301-306. (in Chinese) |
| [10] |
Hodgson E, Lewys-James A, Rao Ravella S, et al. Optimisation of slow-pyrolysis process conditions to maximise char yield and heavy metal adsorption of biochar produced from different feedstocks[J]. Bioresource Technology, 2016, 214: 574-581. DOI:10.1016/j.biortech.2016.05.009 |
| [11] |
Ahmed M B, Zhou J L, Ngo H H, et al. Progress in the preparation and application of modified biochar for improved contaminant removal from water and wastewater[J]. Bioresource Technology, 2016, 214: 836-851. DOI:10.1016/j.biortech.2016.05.057 |
| [12] |
Li S J, Han K H, Li J X, et al. Preparation and characterization of super activated carbon produced from gulfweed by KOH activation[J]. Microporous and Mesoporous Materials, 2017, 243: 291-300. DOI:10.1016/j.micromeso.2017.02.052 |
| [13] |
Syed-Hassan S A S, Zai ni M S M. Optimization of the preparation of activated carbon from palm kernel shell for methane adsorption using Taguchi orthogonal array design[J]. Korean Journal of Chemical Engineering, 2016, 33(8): 2502-2512. DOI:10.1007/s11814-016-0072-z |
| [14] |
肖怀秋, 李玉珍. 微生物培养基-优化方法研究进展[J]. 酿酒科技, 2010(1): 90-94. XIAO Huaiqiu, LI Yuzhen. Research progress in the optimization of microbial culture medium[J]. Liquor-Making Science&Technology, 2010(1): 90-94. (in Chinese) |
| [15] |
Khobragade M U, Nayak A K, Pal A. Application of response surface methodology to evaluate the removal efficiencies of Mn(Ⅱ), Ni(Ⅱ), and Cu(Ⅱ) by surfactant-modified alumina[J]. Clean Technologies Environmental Policy, 2016, 18(4): 1003-1020. DOI:10.1007/s10098-016-1116-0 |
| [16] |
Demim S, Drouiche N, Aouabed A, et al. Study of heavy metal removal from heavy metal mixture using the CCD method[J]. Journal of Industrial and Engineering Chemistry, 2014, 20(2): 512-520. DOI:10.1016/j.jiec.2013.05.010 |
| [17] |
Ghani Z A, Yusoff M S, Zaman N Q, et al. Optimization of preparation conditions for activated carbon from banana pseudo-stem using response surface methodology on removal of color and COD from landfill leachate[J]. Waste Management, 2017, 62: 177-187. DOI:10.1016/j.wasman.2017.02.026 |
| [18] |
Senthilkumar T, Chattopadhyay S K, Miranda L R. Optimization of activated carbon preparation from pomegranate peel(punica granatum peel) using RSM[J]. Chemical Engineering Communications, 2017, 204(2): 238-248. DOI:10.1080/00986445.2016.1262358 |
| [19] |
中国环境监测总站.水质镍的测定丁二酮肟分光光光度法GB 11910-1989[S].北京: 中国环境科学出版社, 1989. China National Environmental Monitoring Centre. Water quality determination of nickel dimethylglyoxime spectrophotometric method GB 11910-1989[S]. Beijing: China Environmental Science Press, 1989. (in Chinese) |
| [20] |
王向前, 胡学玉, 陈窈君, 等. 生物炭及改性生物炭对水环境中重金属的吸附固定作用[J]. 环境工程, 2016, 34(12): 32-37. WANG Xiangqian, HU Xueyu, CHEN Yaojun, et al. Effect of biochar and modified biochar on the adsorption and immobilization of heavy metals in water environment[J]. Environmental Engineering, 2016, 34(12): 32-37. (in Chinese) |
| [21] |
Tan X F, Liu Y G, Zeng G M, et al. Application of biochar for the removal of pollutants from aqueous solutions[J]. Chemosphere, 2015, 125: 70-85. DOI:10.1016/j.chemosphere.2014.12.058 |
| [22] |
Song Z G, Lian F, Yu Z H, et al. Synthesis and characterization of a novel MnOx-loaded biochar and its adsorption properties for Cu2+ in aqueous solution[J]. Chemical Engineering Journal, 2014, 242: 36-42. DOI:10.1016/j.cej.2013.12.061 |
| [23] |
Faheem, Yu H, Liu J, et al. Preparation of MnOx-loaded biochar for Pb2+ removal:Adsorption performance and possible mechanism[J]. Journal of the Taiwan Institute of Chemical Engineers, 2016, 66: 313-320. DOI:10.1016/j.jtice.2016.07.010 |
| [24] |
傅瑞琪, 刘榆, 楼子墨, 等. 氨基改性猪热解炭化物及对水中微量Cu(Ⅱ)和Cu(Ⅱ)-Cit的去除研究[J]. 农业环境科学学报, 2016, 35(10): 1998-2004. FU Ruiqi, LIU Yu, LOU Zimo, et al. Aminized pig biochar and its removals of trace Cu(Ⅱ) and Cu(Ⅱ)-Cit from aqueous solution[J]. Journal of Agro-Environment Science, 2016, 35(10): 1998-2004. (in Chinese) DOI:10.11654/jaes.2016-0428 |
| [25] |
Gan C, Liu Y G, Tan X F, et al. Effect of porous zinc-biochar nanocomposites on Cr(Ⅵ) adsorption from aqueous solution[J]. Rsc Advances, 2015, 5(44): 35107-35115. DOI:10.1039/C5RA04416B |
| [26] |
Inyang M I, Gao B, Yao Y, et al. A review of biochar as a low-cost adsorbent for aqueous heavy metal removal[J]. Critical Reviews in Environmental Science & Technology, 2016, 46(4): 406-433. |
| [27] |
Mukherjee A, Zimmerman A R, Harris W. Surface chemistry variations among a series of laboratory-produced biochars[J]. Geoderma, 2011, 163(3/4): 247-255. |
| [28] |
Lian F, Xing B, Zhu L. Comparative study on composition, structure, and adsorption behavior of activated carbons derived from different synthetic waste polymers[J]. Journal of Colloid & Interface Science, 2011, 360(2): 725-730. |
| [29] |
Xing S T, Hu C, Qu J H, et al. Characterization and reactivity of MnOx supported on mesoporous zirconia for herbicide 2, 4-D mineralization with ozone[J]. Environmental Science & Technology, 2008, 42(9): 3363-3368. |
| [30] |
Yang G X, Jiang H. Amino modification of biochar for enhanced adsorption of copper ions from synthetic wastewater[J]. Water Research, 2014, 48: 396-405. DOI:10.1016/j.watres.2013.09.050 |
| [31] |
Panda G C, Das S K, Bandopadhyay T S, et al. Adsorption of nickel on husk of Lathyrus sativus:Behavior and binding mechanism[J]. Colloids and Surfaces B:Biointerfaces, 2007, 57(2): 135-142. DOI:10.1016/j.colsurfb.2007.01.022 |
| [32] |
Yakout S M. Monitoring the changes of chemical properties of rice straw-derived biochars modified by different oxidizing agents and their adsorptive performance for organics[J]. Bioremediation Journal, 2015, 19(2): 171-182. DOI:10.1080/10889868.2015.1029115 |
 2018, Vol. 41
2018, Vol. 41


